 Use the results in the table to deduce an order of reactivity, starting with the most reactive metal. The table shows the results of one of these investigations. Different combinations of metal and salt solution are tested. A piece of metal is dipped into a salt solution. the blue colour of the solution fades as blue copper sulfate solution is replaced by colourless magnesium sulfate solutionĪ reactivity series can be deduced by carrying out several displacement reactions. 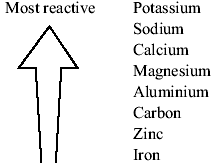
Mg(s) + CuSO 4 (aq) → MgSO 4 (aq) + Cu(s) The following metals are listed in order of reactivity (most reactive first) sodium > magnesium > zinc > copper Describe what each metal does when Reacts with water heated in air added to dilute hydrochloric acid Which of the four metals would be suitable for making saucepans Explain why the others are not. Magnesium + copper sulfate → magnesium sulfate + copper 
It displaces copper from copper sulfate solution : For example, magnesium is more reactive than copper. Displacement in solutionsĪ more reactive metal can displace a less reactive metal from its compounds. At the same time, copper oxide is reduced because oxygen is removed from it. In this reaction, carbon is oxidised because it gains oxygen. For example:Ĭopper oxide + carbon → copper + carbon dioxide explain how the reactivity series is constructed, fill in gaps in the reactivity series, describe the reactions of metals with water, acids, oxygen, hydrogen, and metal oxides, identify the more reactive metal from a pair of metals, relate the reactivity series to the stability of a metal’s compounds. Register free for an online tutoring session to. Oxygen can be removed from metal oxides in chemical reactions. Learn about Reactivity Series Topic of chemistry in detail Explained by subject experts on. The reactions are oxidation reactions because the metal gains oxygen. For example, magnesium burns rapidly in air: 
Many metals react with oxygen to make metal oxides. The worksheet has a focus on aluminium and iron processing techniques, and provides an opportunity to practice completing chemical equations based on the. Oxidation, reduction and displacement reactions Reactions of metals with oxygen 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
